Staff members talk beside the virus culture/inactivation area of a vaccine production plant of China National Pharmaceutical Group (Sinopharm) in Beijing, capital of China, April 10, 2020. China has approved two COVID-19 inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus Tuesday. The two vaccine candidates are developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm) and Sinovac Research and Development Co., Ltd, a company based in Beijing. Clinical trials of the two vaccines have started. (Xinhua/Zhang Yuwei)
BEIJING, April 14 (Xinhua) -- China has approved two COVID-19 inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus Tuesday.
The two vaccine candidates were developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm), and Sinovac Research and Development Co., Ltd, a company based in Beijing. Clinical trials of the two vaccines have started.
The two vaccines are China's first batch of inactivated vaccines for COVID-19 that have obtained clinical trial approval. Using killed pathogenic microorganisms for enhancing the immunogenicity, inactivated vaccines have advantages of mature production process, controllable quality standards and wide protection range.
They can be used for large-scale vaccination, and their safety and effectiveness can be judged by internationally accepted standards.
China has laid a solid foundation for research in inactivated vaccines over the past years. Inactivated vaccines have been widely used to fight hepatitis A, influenza, hand-foot-and-mouth disease and poliomyelitis.
The vaccine developers have the capacity for large-scale production.

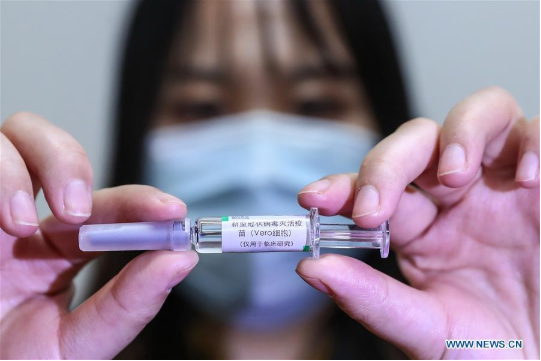
A staff member displays a sample of the COVID-19 inactivated vaccine at a vaccine production plant of China National Pharmaceutical Group (Sinopharm) in Beijing, capital of China, April 10, 2020. China has approved two COVID-19 inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus Tuesday. The two vaccine candidates are developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm) and Sinovac Research and Development Co., Ltd, a company based in Beijing. Clinical trials of the two vaccines have started. (Xinhua/Zhang Yuwei)
According to sources from the Sinopharm, the company has allocated one billion yuan (about 142 million U.S. dollars) for vaccine research and development in two technological approaches. Besides the approved inactivated vaccine, the company is also working on another inactivated vaccine and a genetic engineering vaccine.
Drawing on previous experience in SARS vaccine research and development, the other vaccine developer Sinovac has maintained close cooperation with many institutions, including Zhejiang Provincial Center for Disease Control and Prevention and the Institute of Laboratory Animal Sciences under the Chinese Academy of Medical Sciences.
Sinovac's inactivated vaccine has shown a good cross-neutralization reaction to different COVID-19 strains at home and abroad.
The National Medical Products Administration has also accelerated the approval procedures for COVID-19 vaccines on the premise of their safety and efficacy.
China adopts five technological approaches to develop COVID-19 vaccines, including inactivated vaccines, recombinant protein vaccines, adenovirus vector vaccines, nucleic acid vaccines and vaccines using attenuated influenza viruses as vectors.
The recombinant adenovirus vector vaccine developed by the Institute of Military Medicine under the Academy of Military Sciences started its clinical trial on March 16.
:

Staff members talk beside the virus culture/inactivation area of a vaccine production plant of China National Pharmaceutical Group (Sinopharm) in Beijing, capital of China, April 10, 2020. China has approved two COVID-19 inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus Tuesday. The two vaccine candidates are developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm) and Sinovac Research and Development Co., Ltd, a company based in Beijing. Clinical trials of the two vaccines have started. (Xinhua/Zhang Yuwei)
BEIJING, April 14 (Xinhua) -- China has approved two COVID-19 inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus Tuesday.
The two vaccine candidates were developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm), and Sinovac Research and Development Co., Ltd, a company based in Beijing. Clinical trials of the two vaccines have started.
The two vaccines are China's first batch of inactivated vaccines for COVID-19 that have obtained clinical trial approval. Using killed pathogenic microorganisms for enhancing the immunogenicity, inactivated vaccines have advantages of mature production process, controllable quality standards and wide protection range.
They can be used for large-scale vaccination, and their safety and effectiveness can be judged by internationally accepted standards.
China has laid a solid foundation for research in inactivated vaccines over the past years. Inactivated vaccines have been widely used to fight hepatitis A, influenza, hand-foot-and-mouth disease and poliomyelitis.
The vaccine developers have the capacity for large-scale production.

A staff member displays a sample of the COVID-19 inactivated vaccine at a vaccine production plant of China National Pharmaceutical Group (Sinopharm) in Beijing, capital of China, April 10, 2020. China has approved two COVID-19 inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus Tuesday. The two vaccine candidates are developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm) and Sinovac Research and Development Co., Ltd, a company based in Beijing. Clinical trials of the two vaccines have started. (Xinhua/Zhang Yuwei)
According to sources from the Sinopharm, the company has allocated one billion yuan (about 142 million U.S. dollars) for vaccine research and development in two technological approaches. Besides the approved inactivated vaccine, the company is also working on another inactivated vaccine and a genetic engineering vaccine.
Drawing on previous experience in SARS vaccine research and development, the other vaccine developer Sinovac has maintained close cooperation with many institutions, including Zhejiang Provincial Center for Disease Control and Prevention and the Institute of Laboratory Animal Sciences under the Chinese Academy of Medical Sciences.
Sinovac's inactivated vaccine has shown a good cross-neutralization reaction to different COVID-19 strains at home and abroad.
The National Medical Products Administration has also accelerated the approval procedures for COVID-19 vaccines on the premise of their safety and efficacy.
China adopts five technological approaches to develop COVID-19 vaccines, including inactivated vaccines, recombinant protein vaccines, adenovirus vector vaccines, nucleic acid vaccines and vaccines using attenuated influenza viruses as vectors.
The recombinant adenovirus vector vaccine developed by the Institute of Military Medicine under the Academy of Military Sciences started its clinical trial on March 16.
